 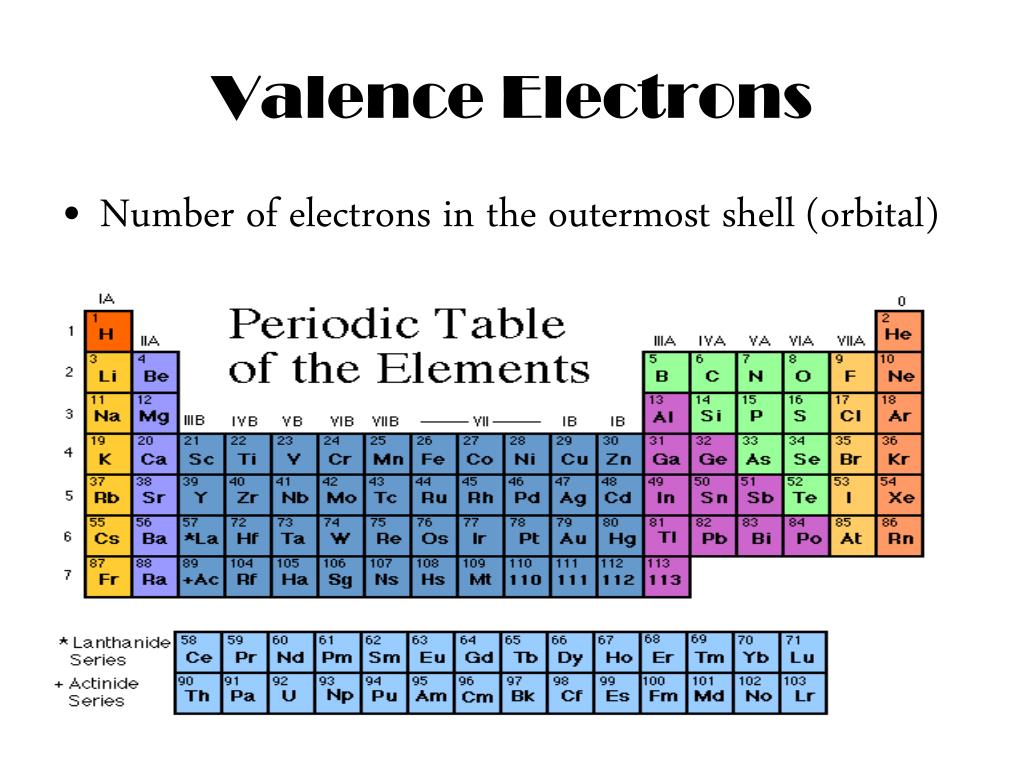  
Hydrogen (H) has atomic number 1, Helium (He) has atomic number 2, etc. Each element in the table has an assigned atomic number.The following notes were taken by me while listening to As another example, an element like chlorine (1s 22s 22p 63s 23p 5) will have three orbital shells: one with two 1s electrons, one with two 2s electrons and six 2p electrons, and one with two 3s electrons and five 3p electrons.Click on any of the squares above for more information on an element.Since the first orbital shell has only two electrons, we know that Boron has two shells: one with two 1s electrons and one with three electrons from the 2s and 2p orbitals. Since its atomic number is five, we know it has five electrons and its electron configuration looks like this: 1s 22s 22p 1. For example, let's say we're looking at the element Boron (B).Besides the very first shell, which can hold only two electrons, each shell can have eight electrons (except, again, when dealing with transition metals.) This is called the Octet Rule. When we're dealing with atoms outside of the transition metals, we say that these orbitals form "orbital shells" around the nucleus, with each successive shell being further out than the ones before. As electrons are added to an atom, they fall into various orbitals according to the order given above - the first two go into the 1s orbital, the two after that go into the 2s orbital, the six after that go into the 2p orbital, and so on. For more on electron configurations, see also this article.Īssign electrons to orbital shells with the Octet Rule.You only need to change the number in the final orbital - the rest is the same since the orbitals before the final one are completely full. Notice that the number of electrons adds up to 17: 2 + 2 + 6 + 2 + 5 = 17.For example, if we want to make the orbital diagram for chlorine (Cl), element 17, which has 17 electrons, we would do it like this: Now that you have this, all you need to do to find another atom's electron configuration is just fill in this pattern from the beginning until you run out of electrons.It has the most electrons of any element, so its electron configuration demonstrates all of the possibilities you could encounter in other elements:ġs 22s 22p 63s 23p 64s 23d 104p 65s 24d 105p 66s 24f 145d 106p 67s 25f 146d 107p 6 
Examine complete electron configuration for oganesson (Og), element 118, which is the last element on the periodic table.If you have to find it yourself, see below: X Research source Once you know an element's electron configuration, finding its number of valence electrons is quite simple (except, of course, for the transition metals.) If you're given the configuration from the get-go, you can skip to the next step. Their electron capacities are as follows:įind the electron configuration for the element you are examining. Keep in mind that each subshell has a certain electron capacity.That's 11 electrons total - sodium is element number 11, so this makes sense. So, for our example, we would say that sodium has 2 electrons in the 1s orbital plus 2 electrons in the 2s orbital plus 6 electrons in the 2p orbital plus 1 electron in the 3s orbital.The (number)(letter) chunk is the name of the electron orbital and the (raised number) is the number of electrons in that orbital - that's it! (number)(letter) (raised number)(number)(letter) (raised number). Notice that this electron configuration is just a repeating string that goes like this:.Let's look at an example configuration for the element sodium (Na):.These may at first look complicated, but they're just a way to represent the electron orbitals in an atom with letters and numbers and they're easy once you know what you're looking at. Another way to find an element's valence electrons is with something called an electron configuration. Learn how to read an electron configuration. This means that an atom can have multiple numbers of valence electrons depending on how it is manipulated. For reasons that are a little too complex to explain here, when electrons are added to the outermost d shell of a transition metal (more on this below), the first electrons that go into the shell tend to act like normal valence electrons, but after that, they don't, and electrons from other orbital layers sometimes act as valence electrons instead.Generally, the valence electrons are the electrons in the outermost shell - in other words, the last electrons added. As electrons are added to an atom, they are sorted into different "orbitals" - basically different areas around the nucleus that the electrons congregate in.See below for a quick run-through or skip this step to get right to the answers. Understanding why transition metals don't really "work" like the rest of the periodic table requires a little explanation of the way electrons behave in atoms. Understand that transition metals don't have "traditional" valence electrons. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
